Clinical and Economic Impact
of CAUTIs and the Role of
Contactless Monitoring.
Meta Description:
atheter-associated urinary tract infections (CAUTIs) are among the most common healthcare-associated infections and are…
Introduction
Catheter-associated urinary tract infections (CAUTIs) are among the most common healthcare-associated infections and are a particular concern in intensive care units (ICUs). A CAUTI occurs when bacteria enter the urinary tract via an indwelling catheter and cause infection. Because many ICU patients require indwelling urinary catheters for hourly urine output monitoring or other reasons, CAUTIs occur disproportionately in high-acuity settings. They not only cause patient discomfort and morbidity but can lead to serious complications like urosepsis and prolonged hospital stays. This article examines the clinical and economic impact of CAUTIs in ICUs, discusses how current practices (such as manual urine output monitoring) contribute to the problem, and explores how contactless, automated urine monitoring systems (such as Exypnos) offer a safer, more reliable alternative.
CAUTI Prevalence and Clinical Burden in ICUs
Urinary tract infections are a leading type of hospital-acquired infection, accounting for an estimated 62,700 cases in U.S. acute care hospitals in 2015. Importantly, virtually all healthcare-associated UTIs are linked to urinary catheters. In ICUs the problem is amplified: at any time, 60–90% of ICU patients may have a catheter in place (compared to 10–30% on general wards), and nearly 95% of UTIs in ICU settings are catheter-associated. Infection surveillance data show ICU CAUTI incidence rates on the order of 3–7 infections per 1,000 catheter-days – several-fold higher than in lower-acuity units. Each day of catheterization poses a compounding risk (roughly a 3–7% increase in infection risk per day of indwelling time), so extended catheter use greatly elevates the chance of infection.
The clinical burden of CAUTIs in critically ill patients is substantial. A bladder infection in a fragile ICU patient can quickly escalate: CAUTIs frequently lead to complications such as pyelonephritis or gram-negative bacteremia. Indeed, CAUTIs are a leading cause of secondary sepsis in the hospital – roughly 17% of hospital-acquired bacteremias are attributed to a urinary source. If a urinary infection progresses to bloodstream infection in an ICU patient, mortality rates are high (on the order of 10–30% in reported series). Even when not causing overt sepsis, CAUTIs prolong ICU and hospital length of stay and necessitate invasive procedures (e.g. central lines for antibiotics) that can trigger further complications. The U.S. CDC estimated that over 13,000 deaths each year are associated with UTIs, mostly catheter-related. In short, CAUTIs add significant morbidity and mortality to already high-risk ICU patients, making prevention a top priority.
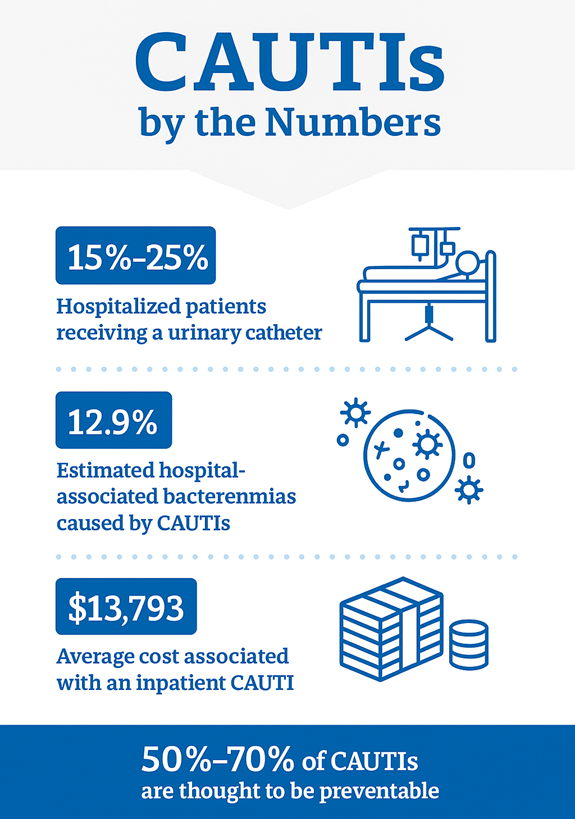
Economic Impact of CAUTIs
In addition to clinical harm, CAUTIs carry a heavy economic impact. Treating a single catheter-associated UTI involves extra laboratory tests, antimicrobial therapy, and often days of additional in-hospital care. Estimated costs per CAUTI case vary, but studies peg the average inpatient cost in the range of $1,000 up to $13,000 or more. Notably, ICU-acquired CAUTIs tend to be the most expensive, since these patients are often sicker and the infections more severe (one analysis found an average cost of about $10,000 for a CAUTI in ICU patients). Across the United States, the aggregate cost burden is hundreds of millions of dollars annually. These infections also incur indirect costs: they can extend ICU occupancy (blocking new admissions), require costly isolation or enhanced nursing care, and contribute to antimicrobial resistance through antibiotic use.
Because CAUTIs are considered largely preventable, payers and regulators have taken note. In the U.S., the Centers for Medicare & Medicaid Services (CMS) identifies CAUTI as a preventable hospital-acquired condition and may deny hospitals reimbursement for its treatment. This creates financial incentives for hospitals to reduce CAUTI rates. Additionally, an avoidable infection adversely affects quality metrics and can lead to financial penalties under value-based purchasing programs. In summary, every CAUTI in the ICU not only endangers the patient but also carries a price tag – one that hospitals increasingly must absorb, providing strong motivation to invest in better prevention and monitoring strategies.

Why Manual Urine Output Monitoring Contributes to Risk
One less-appreciated factor in CAUTI risk is the traditional method of monitoring urine output in ICUs. Accurate urine output measurement is vital for managing critically ill patients (for example, tracking hourly output is key for guiding fluid resuscitation and detecting renal impairment). This need for precision is a major reason so many ICU patients are catheterized. However, manual urine output monitoring introduces infection risks and can delay recognition of patient deterioration.
From an infection control standpoint, each interaction with the catheter or drainage bag is a potential entry point for bacteria. Best practices dictate maintaining a closed sterile drainage system continuously, because opening or disconnecting the system even briefly allows pathogens to enter. Studies have shown that if a urinary drainage system is left open to air (no closed circuit), the infection rate approaches 100%, whereas using a continuously closed system can keep infection rates under 25%. Consequently, frequently breaking the catheter system to empty urine or measure output will increase CAUTI risk. In many ICUs, nurses empty urometer chambers or bags hourly to record output, and each of those manipulations – even done with gloves and aseptic technique – is an opportunity for contamination.
Equally important, manual monitoring is prone to lapses in timeliness and accuracy, which can result in missed warning signs like oliguria. ICU nurses typically document urine output hourly, but in practice these readings often run behind or get batched if the unit is busy. In a study comparing electronic monitoring to nurse charting, manual urine outputs were recorded with an average delay of 47 minutes (median ~18 min), and occasionally up to 6 hours late. Nurses also tended to overestimate volumes by around 20 mL per hour compared to the actual output captured by an automated device. These delays and inaccuracies mean a drop in urine output might go unnoticed for hours. A patient could develop oliguria (low output) at 2 PM, but if staff are tied up with an emergency, it may not be measured and recognized until much later. This matters because prolonged unrecognized oliguria is dangerous – one analysis found that a delay of more than 6 hours in addressing oliguria was associated with a ~30% increase in ICU mortality risk. In summary, traditional urine monitoring forces a trade-off: checking output frequently is clinically important but doing so manually increases infection exposure and is limited by human timing. This is where automation offers a compelling solution.
Contactless, Automated Urine Monitoring: A Safer Alternative
New “contactless” automated urine monitors have been developed to mitigate the pitfalls of manual monitoring. These systems allow continuous urine output measurement without requiring hourly human intervention or any opening of the drainage system. For example, the Exypnos urine monitoring system uses an optical sensor and a closed, inline collection chamber to automatically measure urine volume in real time. The disposable chamber connects seamlessly to standard Foley catheter tubing and remains sealed, so urine flows through a sterile pathway that the device can measure without ever exposing the urine to the environment. Urine volume data are transmitted continuously to a bedside monitor or central station, giving up-to-the-minute insight into output.
Because the sensor does not require breaking the circuit or touching the urine, the closed sterile drainage is maintained at all times, dramatically reducing contamination risk. In essence, automated monitors uphold the “closed system” ideal far better than any manual method. There is no need for a nurse to open a drain port every hour – the device does the monitoring automatically. Additionally, these systems are highly accurate. In the ICU study mentioned earlier, the electronic monitor’s measurements were virtually identical to true output (within a couple milliliters), whereas nurse measurements showed significant error. This means clinicians can trust the readings and not worry about charting delays or misestimation. If a patient’s urine output drops suddenly, the change is evident immediately on the monitor, and alarms can be set to alert staff promptly.
Clinical Advantages Beyond Infection Control
Implementing contactless automated urine monitoring in ICUs provides benefits that extend beyond reducing CAUTIs. One major advantage is earlier detection of acute kidney injury (AKI) and other deterioration. Urine output is a sensitive indicator of renal perfusion – often declining hours or days before serum creatinine levels rise. Studies show that relying on creatinine alone can miss a large portion of AKI in critical care; in one analysis, serum creatinine criteria failed to identify over one-third of AKI cases that were detectable by oliguria. Moreover, even brief episodes of oliguria are prognostically significant: ICU patients who meet AKI criteria by urine output (oliguria) have been shown to have higher mortality than those who meet it by creatinine rise alone. By delivering continuous urine output trends, automated monitors enable the care team to recognize AKI in its earliest phase and intervene sooner (for example, by optimizing volume status, avoiding nephrotoxic drugs, or addressing causes of low output). This proactive approach could improve renal outcomes and overall patient survival.
Another benefit is enhanced fluid and hemodynamic management. Minute-to-minute urine output is essentially a real-time gauge of kidney perfusion and volume status. With continuous monitoring, clinicians can respond faster to a patient’s fluid needs – for instance, detecting an insidious drop in output that might signal the onset of shock or, conversely, noticing a brisk diuresis that could lead to dehydration. Research indicates that patients who undergo more intensive urine output monitoring tend to have less fluid overload and better outcomes than those monitored less frequently. Automated systems make such intensive monitoring feasible around the clock without increasing staff workload.
Finally, there are workflow and safety advantages for nursing staff. Automation frees ICU nurses from the repetitive task of measuring and emptying urine bags every hour. This not only saves time (allowing nurses to focus on direct patient care) but also reduces staff exposure to potentially infectious urine. Early implementations have shown that automated urine monitoring can streamline nursing workflow and reduce time spent on documentation and waste handling. In high-acuity environments, where nurse-to-patient ratios are tight, this efficiency gain is very valuable. Additionally, the continuous data logs can be integrated into electronic medical records, improving chart accuracy and enabling advanced clinical decision support (such as alerting if output falls below a set threshold).
Conclusion
CAUTIs impose a serious clinical and financial burden in ICUs, but innovations in urine output monitoring are helping turn the tide. By eliminating unnecessary breaks in the catheter closed system and by providing immediate awareness of changes in renal output, contactless automated monitoring systems directly tackle two key contributors to CAUTIs: contamination during manual measurements and delayed recognition of patient decline. Early studies and real-world use indicate that these technologies can reduce infection risk, enhance early AKI detection, optimize fluid management, and ease the workload on ICU staff – all without compromising the quality of patient data. For intensivists and infection control teams, such systems represent a promising strategy to improve ICU care. In an era where hospitals are increasingly accountable for preventing device-related infections, automated urine monitoring offers a compelling, evidence-based tool to improve patient safety and outcomes in high-acuity environments.
Sources:
- Centers for Disease Control and Prevention (CDC). Clinical Safety: Preventing Catheter-associated Urinary Tract Infections (CAUTIs). June 27, 2025.
- NHSN Patient Safety Component Manual: Urinary Tract Infection Events. January 2025
- California Department of Public Health (CDPH). CAUTI Prevention Guidelines. 2017
- Nicastri E, Leone S. Healthcare Associated Urinary Tract Infections – ISID Guide to Infection Control. June 2021
- CDC/HICPAC. Guideline for Prevention of CAUTI. 2009
- Cho HJ, et al. Impact of Infection Prevention Programs on CAUTI in ICUs. J Korean Med Sci. 2024
- NHSN Data: “UTIs are 5th most common HAI” (2015 data)
- E, et al. Health Care–Associated Infections: A Meta-analysis of Costs. JAMA Intern Med. 2013
- Additional Hospital Inpatient Cost of CAUTI – Meta-analysis. 2017
- ISID Guide (Nicastri & Leone). CAUTI costs and outcomes.
- Meddings J, et al. Effect of Nonpayment for Hospital CAUTIs. Ann Intern Med. 2012
- Smith A, Kellum JA, et al. Automated vs Manual Urine Output Monitoring in ICU. Crit Care. 2021
- Smith A, Kellum JA, et al. Urine Output and AKI Detection in ICU. Crit Care. 2021
- Jin K, et al. Intensive UO Monitoring and Outcomes. (Referenced in Smith et al. 2021)
- Exypnos Medical. Product Overview: Exypnos UO Monitoring System. 2025
- Exypnos Medical. Clinical Impact of Manual vs Automated Monitoring. 2025
- Exypnos Business Plan (user-provided data). Nursing Workflow Benefits. 2025
